Characterization of calcium oxide catalysts from natural sources and their application in the transesterification of sunflower oil - ScienceDirect

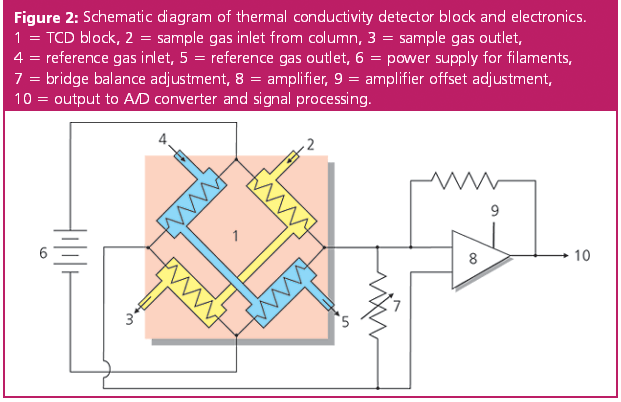
GC-TCD spectra of (a) N 2 , (b) H 2 and (c) the gas released from the... | Download Scientific Diagram

Matrix Effects in a Fluid Catalytic Cracking Catalyst Particle: Influence on Structure, Acidity, and Accessibility - Velthoen - 2020 - Chemistry – A European Journal - Wiley Online Library
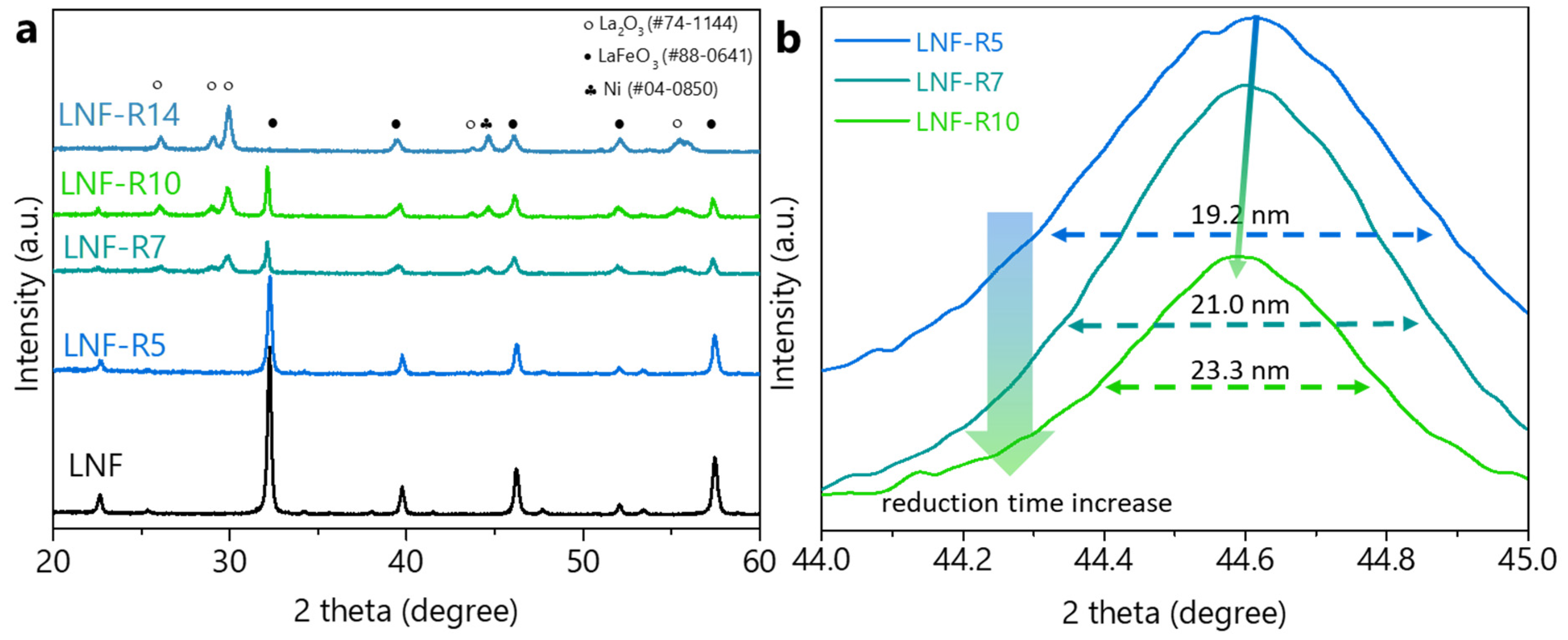
ChemEngineering | Free Full-Text | Improved Catalytic Activity of the High-Temperature Water Gas Shift Reaction on Metal-Exsolved La0.9Ni0.05Fe0.95O3 by Controlling Reduction Time

Uniform Pt/Pd Bimetallic Nanocrystals Demonstrate Platinum Effect on Palladium Methane Combustion Activity and Stability | ACS Catalysis

Electrochemical Production of Sustainable Hydrocarbon Fuels from CO2 Co-electrolysis in Eutectic Molten Melts | ACS Sustainable Chemistry & Engineering

Selective Catalytic Reduction of NOx by Methanol on Metal-Free Zeolite with Brønsted and Lewis Acid Pair | ACS Catalysis

Sustainable Conversion of Carbon Dioxide into Diverse Hydrocarbon Fuels via Molten Salt Electrolysis | ACS Sustainable Chemistry & Engineering

Accurate Evaluation of Active-Site Density (SD) and Turnover Frequency (TOF) of PGM-Free Metal–Nitrogen-Doped Carbon (MNC) Electrocatalysts using CO Cryo Adsorption | ACS Catalysis

Upgrading syngas from wood gasification through steam reforming of tars over highly active Ni-perovskite catalysts at relatively low temperature - ScienceDirect

Effect of Defects on Optical, Electronic, and Interface Properties of NiO/SnO2 Heterostructures: Dual-Functional Solar Photocatalytic H2 Production and RhB Degradation | ACS Applied Materials & Interfaces

A comparative study of hydrothermal aging effect on cerium and lanthanum doped Cu/SSZ-13 catalysts for NH3-SCR - ScienceDirect