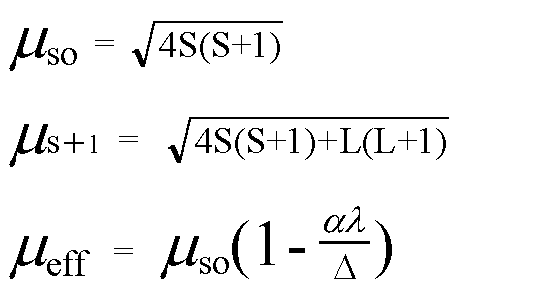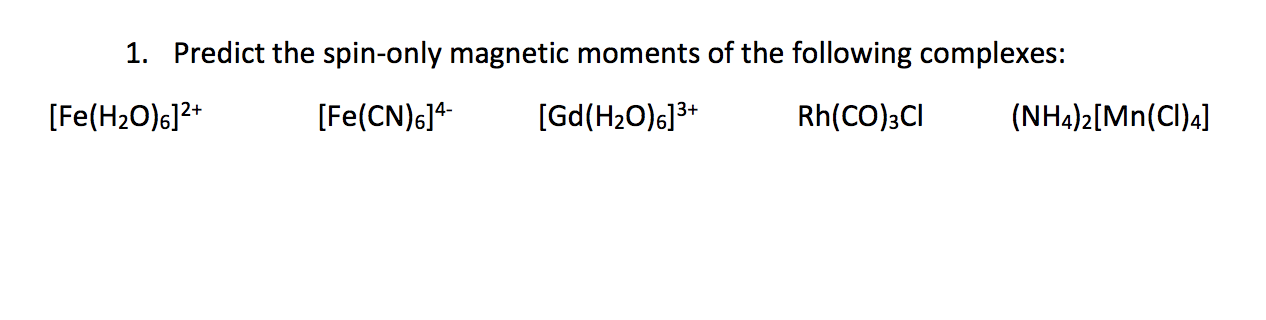![The spin-only magnetic moments of [Mn(CN)(6)]^(4-) and [MnBr(4)]^(2-) in Bohr Magnetons, respectively, are The spin-only magnetic moments of [Mn(CN)(6)]^(4-) and [MnBr(4)]^(2-) in Bohr Magnetons, respectively, are](https://d10lpgp6xz60nq.cloudfront.net/ss/web/618499.jpg)
The spin-only magnetic moments of [Mn(CN)(6)]^(4-) and [MnBr(4)]^(2-) in Bohr Magnetons, respectively, are
![The spin-only magnetic moment of `[Fe(NH_(3))_(6)]^(3+) and [FeF_(6)]^(3-)` (in units of BM ) - YouTube The spin-only magnetic moment of `[Fe(NH_(3))_(6)]^(3+) and [FeF_(6)]^(3-)` (in units of BM ) - YouTube](https://i.ytimg.com/vi/KVj56QvOV1I/maxresdefault.jpg)
The spin-only magnetic moment of `[Fe(NH_(3))_(6)]^(3+) and [FeF_(6)]^(3-)` (in units of BM ) - YouTube

Spin only magnetic moment of `._(25)Mn^(x+)`ion is `sqrt(15)` B.M. Then, the value of `x` is `:` - YouTube

SOLVED: Calculate the magnetic moment of V 3+, Cr3+, Pr3+, Nd3+ according to the following instructions. (a) Consider spin-only magnetic moment in your calculation (b) Consider both spin and orbital moment in
The value of the 'spin only' magnetic moment for one of the following configurations is 2.84BM. The correct one is - Sarthaks eConnect | Largest Online Education Community

The spin only magnetic moment of a divalent ion in aqueous solution (atomic number 29) is ______ BM. Option: 1 2 Option: 2 - Option: 3 - Option: 4 -

The highest value of the calculated spin-only magnetic moment (in BM) among all the transition - YouTube